WHAT is the biggest threat to our way of life or even our continued existence?
Climate change? Economic meltdown? Nuclear armageddon?
They are all very real dangers.
But there is another threat that really brings doctors out in a sweat: antibiotic resistance.
It may not sound very frightening. Not in the way nuclear war does. But the increasing ineffectiveness of antibiotics against the bacteria that cause infections is, in its way, just as much of a threat as any of those others.
Dr Neil Todd certainly believes so. "I'm extremely worried," admits the microbiologist and clinical director of laboratory medicine at York Hospitals Trust. "I don't think it is overblown at all to compare it to the risk of global warming or other large scale problems."
We have relied on antibiotics to control bacterial infections ever since Sir Alexander Fleming discovered penicillin - quite by accident - in 1928.

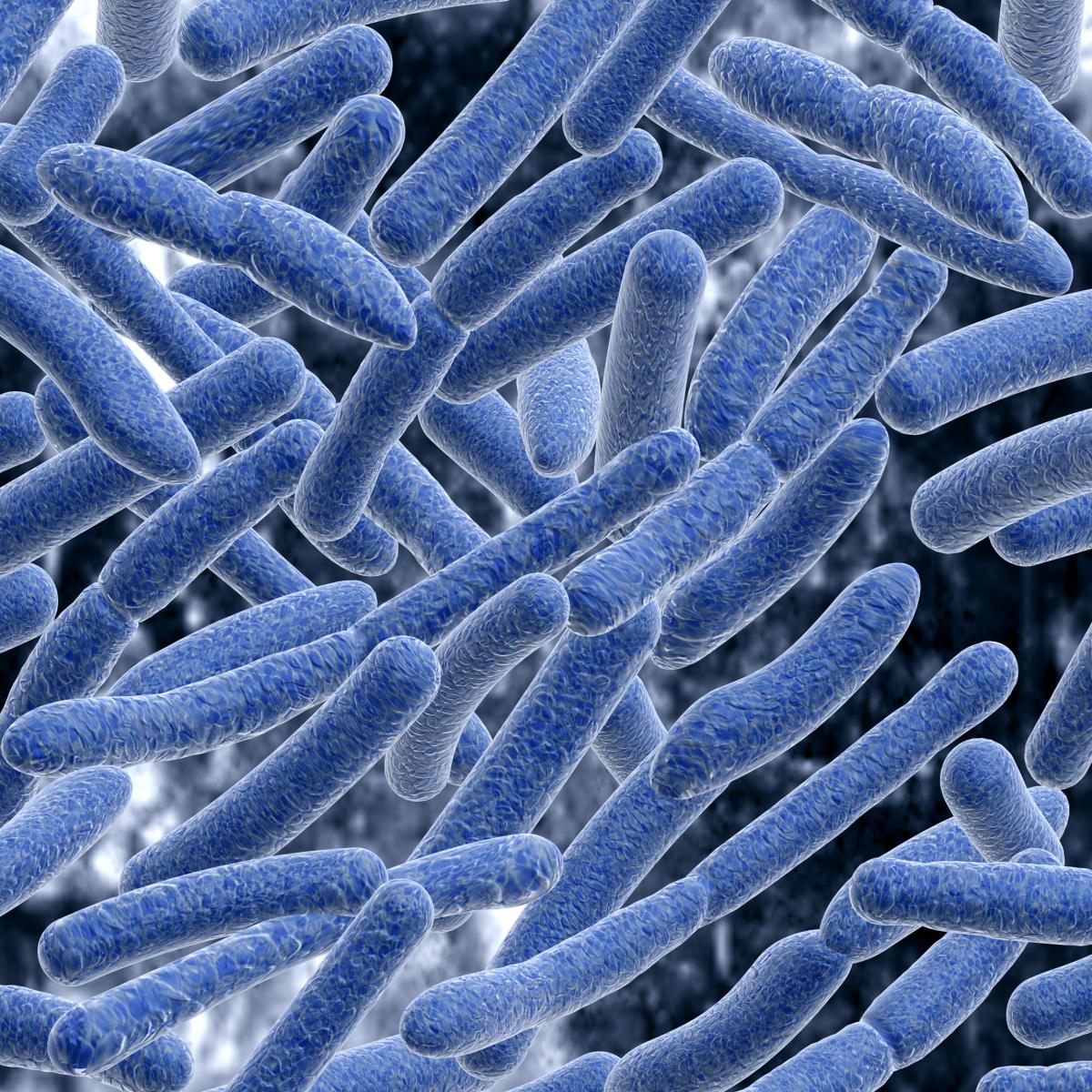
Escherichia coli, a common gut bacterium that can become antibiotic resistant
But increasingly over the last 10-15 years, strains of bacteria have been evolving which are resistant to antibiotics, and which cannot be killed by them. We've all heard of MRSA. But that's just one - and not the most serious - of the new, resistant strains of bacteria that have emerged.
Hospitals and health services across the country and around the world are struggling to cope.
Simple infections, when caused by a resistant strain of bacteria, can spread very quickly - and can be very hard to clear up. "This is causing enormous problems for health services globally," says Dr Todd.

Dr Neil Todd
Patients with infections caused by antibiotic-resistant bacteria have to be isolated in side rooms. Just as with deadly Ebola virus, the staff that treat patients have to wear protective clothing, both to prevent spread of the infection, and to make sure they don't pick it up themselves.
Patients with resistant infections tend to be treated with 'broad-spectrum' antibiotics - which can cause unpleasant side effects such as diarrhoea. And even so, it can take much longer than usual for such infections to be brought under control - so patients stay in hospital longer than they would otherwise need.
Antibiotic-resistant infections can also lead to what Dr Todd calls 'treatment failure'. Infections may not clear up at all, but simply keep getting worse. The patient may develop abscesses. Skins grafts may fail to take. And ultimately patients can die.
All these things are happening already. According to the statistics, something like 5,000 people die in the UK every year from antibiotic-resistant infections, says Prof Colin Garner, the York-based cancer researcher and founder of York Against Cancer, who is now heading up a new national charity based in York dedicated to developing new antibiotics - and finding new ways of using the ones we already have. If anything, the statistics underestimate the problem, he says. Many more people each year die from pneumonia or blood-poisoning, in which the underlying case is an antibiotic-resistant infection.

Prof Colin Garner
To understand why doctors are so worried, you only have to try to imagine what the world would be like if antibiotics failed completely - ie if all bacteria became resistant to them.
Modern medicine as we know it would no longer be possible, says Prof Garner. Simple operations - hip replacements or even tooth extractions - would become much more risky, because of the danger of infection. Transplants would become almost impossible. Cancer treatments, which often suppress the body's immune response, could lead to catastrophic infections and death. Even childbirth would become enormously more risky.
A simple scratch picked up in the garden could cause you to die from an infection. And often, doctors might have to resort to amputations to stop infection spreading - the way they did in the First World War. Of the ten million soldiers who died in that war, about one third died from infections, because we didn't, in those days, have the antibiotics to control them.

Staphylococcus aureus bacteria
We wouldn't be in quite such a bad position in a future, 'post-antibiotic' world, admits Dr Todd, because medicine has developed in other ways. We have better hygiene, better drugs, better vaccines than we did 100 years ago. But even so, a world in which we could no longer use antibiotics would be grim.
So how have we got here?
It essentially comes down to a classic Darwinian struggle for survival by the bacteria themselves, says Dr Todd.
Bacteria, like most other life forms on the planet, evolve. When they encounter something that threatens them - such as an antibiotic - many of them are quickly killed off. But if some have a mutated gene that protects them against the antibiotic, they will survive.
As a result, antibiotic resistance can spread incredibly quickly amongst bacteria. They are very good at swapping genes with each-other, Dr Todd says, so that a gene that confers resistance spreads rapidly from bacterium to bacterium. Bacteria also reproduce quickly. "A bacterium can reproduce every 20 minutes, whereas it takes us 20 years." So bacteria that carry the resistance gene quickly pass it on to the next generation of bacteria. So quickly, that you can sometimes see new, resistant strains springing up in a matter of a few hours or days.
Our widespread over-use of antibiotics - in situations where we'd often be better off allowing our bodies' own defences to heal us - has added to the problem. Exposing bacteria to antibiotics speeds up the rate at which they develop resistance, Dr Todd says: so the more we use antibiotics, the quicker bacteria become resistant to them. Non-resistant strains are killed off - allowing the resistant strains to rapidly spread and take over.

Image of microscopic bacteria glowing green
The rapid evolution of bacterial resistance to antibiotics is made worse because we haven't really developed any new antibiotics for 30 years, Prof Garner says. We have effectively taken our eye off the ball in the arms race against bacteria.
Which is where Prof Garner and the York-based charity he heads - Antibiotic Resarch UK, or ANTRUK for short - comes in.
They have come up with a deceptively simple idea that should enable us to make much better use of the wide range of antibiotics we already have.
These have been becoming steadily less effective as new, resistant strains of bacteria evolve.
But there is evidence that suggests that by using them in combination with other drugs, it may be possible to break down the bacteria's resistance, so that the antibiotics work again.
ANTRUK plans to test common antibiotics in combination with more than 2,000 other, wide-ranging drugs and medicines in common use - medicines as varied as aspirin and imodium - to see if the combination restores the antibiotic's ability to kill bacteria.

Prof Colin Garner
They will be screening the drugs one by one, identifying combinations that look promising for further testing.
Prof Garner estimates it could take as little as four to six months and £250,000 to complete the initial screening.
Promising combinations of drug and antibiotic would then be taken forward for further clinical development - the next stage in preparing them ready for use - before ultimately being used in clinical trials on patients.
The aim would be to have the first combination treatment available by the early 2020s, he says - with other combinations being developed thereafter.
We will still need to develop completely new antibiotics, if we are to regain the initiative in the war against bacteria, Prof Garner says. ANTRUK hopes that one day, with the right funding, it may be able to play a part in that.
But it could take 15-20 years and many millions of pounds to develop a new antibiotic.
In the meantime, ANTRUK's approach of trying to develop new treatments based on using existing antibiotics in combination with other common medicines should help bridge the gap until those new antibiotics are ready.
"This will buy us some time," Prof Garner says.
What is ANTRUK?
Antibiotic Research UK (ANTRUK) is a national charity set up a year ago. Based in York, it includes leading British university and pharma company scientists and clinicians.
Prof Garner, the charity's chief executive, headed up a cancer research laboratory in York years, and was one of the founders of York Against Cancer.
ANTRUK has already raised £110,000 to cover its core costs and is now seeking the £250,000 to screen more than 2,000 drugs in combination with antibiotics, to see if the combination is effective at killing bacteria. The charity estimates it will then need between £2-£5 million to take promising combinations of drugs and antibiotics forward for clinical development. Over the next 5-7 years, it aims to raise about £30 million to bring at least one new antibiotic therapy to market.
It sounds a lot of money. But compared to the cost of developing completely new drugs, it is actually not much at all.
To find out more about ANTRUK, visit www.antibioticresearch.org.uk
The war against bacteria
Bacteria are tiny, single-celled organisms protected by a tough cell wall. The type of bacteria ANTRUK will focus on are the so-called 'gram-negative' bacteria. These have a double-shell, consisting of an inner cell wall surrounded by an outer membrane. It is this double shell that makes them resistant to antibiotics.

3D representation of a bacteria cell wall
Antibiotics often work by attaching themselves to the cell wall of the bacterium. The outer membrane of gram-negative bacteria prevents antibiotics reaching the inner cell wall hidden inside - so that they no longer work.
What is needed is something to help the antibiotics puncture that outer membrane, a bit like puncturing a football, Prof Garner says. There is evidence that some commonly-used drugs, when used in combination with antibiotics, will help do this.
The need for new antibiotic therapies is urgent.
The Chief Medical Officer, Dame Sally Davies, has described antibiotic resistance as a 'catastrophic threat'. "If we don’t act now, any one of us could go into hospital in 20 years for minor surgery and die because of an ordinary infection that can’t be treated by antibiotics," she said.
In a bid to reduce the over-use of antibiotics when they are not needed - a practice blamed for helping to make bacteria more resistant - the National Institute for Health and Care Excellence (NICE) recently published guidance for GPs, nurses and pharmacists, urging them to resist pressure from patients to prescribe antibiotics when not needed.
NICE says as many as 10 million antibiotic prescriptions are dished out unnecessarily every year.
Often patients want antibiotics for common cough and colds, Prof Garner says - conditions usually caused by viruses, against which antibiotics don't work.



Comments: Our rules
We want our comments to be a lively and valuable part of our community - a place where readers can debate and engage with the most important local issues. The ability to comment on our stories is a privilege, not a right, however, and that privilege may be withdrawn if it is abused or misused.
Please report any comments that break our rules.
Read the rules hereLast Updated:
Report this comment Cancel